Bài viết nổi bật, Tin tức chung, Tin tức sinh viên, Tuyển dụng
THÔNG BÁO VỀ NỘP HỒ SƠ HỌC BỔNG DU HỌC TIẾN SỸ TẠI UNIVERSITY OF SYDNEY (Nộp trước ngày 20/08)
- THÔNG TIN HỌC BỔNG:
Thông tin học bổng chi tiết ở link bên dưới:
https://cerebralpalsy.org.au/research-foundation/phd-grant/
Một số điểm lưu ý khác không ghi trong thông tin học bổng:
- Nếu thí sinh được học bổng (tối đa 35,000 AUD/năm) và đăng ký học tiến sĩ tại Đại học Sydney thì tiền học (tuition fee) sẽ được miễn phí và số tiền trên sẽ sử dụng như chi phí hàng tháng.
- Chương trình Tiến sĩ tại Đại học Sydney thông thường kéo dài 3-4 năm và học bổng kéo dài tối đa 3 năm, trong trường hợp thí sinh học 4 năm thì năm cuối Đại học Sydney có nhiều chương trình học bổng dành riêng cho năm cuối để hoàn thành chương trình.
- ĐỐI TƯỢNG HỌC BỔNG:
Dành cho các bạn kỹ thuật (Engineering) mong muốn làm nghiên cứu về lĩnh vực Kỹ thuật Y sinh (Biomedical Engineering).
Hướng nghiên cứu: Hiện đang có 2 project đang mở được mô tả kỹ hơn ở các trang tiếp theo:
- Phát triển hệ thống mũ chụp đo kết hợp EEG và eye tracker để theo dõi phát triển vận động thần kinh của trẻ Bại Não trong quá trình tập phục hồi chức năng.
- Phát triển và tích hợp hệ thống hiệu chỉnh cử động trong chụp MRI não bộ, sử dụng trong chụp hoạt động thần kinh của trẻ Bại Não
Các kỹ năng có thể áp dụng cho nghiên cứu: điện, điện tử, lập trình nhúng, AI, robotic, xử lý tín hiệu, xử lý hình ảnh.
Người hướng dẫn: Prof. Alistair McEwan, Đại học Sydney và Tổ chức hỗ trợ Bại Não Australia
https://www.sydney.edu.au/engineering/about/our-people/academic-staff/alistair-mcewan.html
III. YÊU CẦU HỒ SƠ:
Do học bổng sẽ đóng sau ngày 31/08/2023, yêu cầu nộp hồ sơ sơ lược cho TS. Nguyễn Minh Đức, email: duc.nguyenminh5@hust.edu.vn hoặc minh.d.nguyen@sydney.edu.au, trước ngày 20/08.
Hồ sơ yêu cầu:
- Bảng điểm:
- Yêu cầu bằng giỏi trở lên (>3.2 GPA), tính đến trước khi bảo vệ dành cho các bạn sắp ra trường.
- Có thể in ra từ hệ thống, không nhất thiết bảng điểm cứng nếu chưa có.
- Một văn bản liệt kê các thành tích khác như bài báo, thành tích khoa học, thành tích hoạt động xã hội liên quan hay bất kỳ thành tích nào bản thân thấy đáng nói đến.
- Một văn bản liệt kê chi tiết các project đã làm trong quá trình học tại Bách Khoa bao gồm: tên project, tóm tắt mục tiêu và kết quả, và nêu rõ các kỹ năng nào đã áp dụng.
PROJECT PROPOSALS FOR CEREBRAL PALSY RESEARCH
Hiện có hai project đang cần các bạn nghiên cứu sinh tham gia để thực hiện, trong đó project thứ 2 (làm về MRI) cấp thiết hơn và được ưu tiên hơn.
- DESIGNING A MULTIMODAL CAP TO MONITOR NEURODEVELOPMENT
- Backgrounds
Children with CP suffer from impaired neuromotor causing numerous issues such as stiff muscles, reduced movement range, hypertonicity, spasticity, etc. Rehabilitation is the key and is recommended to start as early as 6 months old. Until the age of four, the neuroplasticity, i.e. the capability of a person’s brain to strengthen or rewire neuron networks when learning or refining new actions or knowledge, is enormous. This is the critical window for children’s brains to learn very effectively new sets of movements for the affected limbs. To maximise neuroplasticity during this period, children’s brain progressing through different plays and activities needs to be monitored rigorously to guarantee their playing styles and exercises produce positive brain changes for the affected limbs. However, the only current solution to monitor a child’s neuromotor development is clinical assessment tools, which are too coarse to detect a small but vital change after one rehabilitation program. Furthermore, Only pediatricians/physiotherapists with a license to use the tools can assess, which limit the capability to continuously assess the children’s neurodevelopment. Therefore, there is an enormous need for a solution to monitor children’s brain development progress automatically and continuously that can be used either at home or a rehab centre.
- Proposal
We will design an infant suitable cap that contained a set of suitable wireless physiological sensors (Figure 1). This will be the first reconfigurable wearable that combines technologies including brain signal monitoring (EEG), eyegaze and physiological measurements in a single headcap suitable for infants and reconfigurable for different ages and settings. Our preliminary work in adults identified and solved issues with:
(a) transfer of EEG electrodes to enable simultaneously wearing an eyegaze device,
(b) synchronisation between different sensor systems,
(c) differences in power spectral densities and phase quantities due to different signal processing pipelines for EEG signals adapting signal processing for personalised EEG signals
(d) the presence of bulky wired backpack based physiological monitors not being acceptable to some children and families.
We will address these issues through design of a system based on a single printed circuit board to handle all physiological signals including EEG, eyegaze and movement.
The proposed cap has to satisfy three characteristics: (1) completely wearable to promote maximal comfort but still be able to achieve good SNR, (2) fully optimised toward small children under age of 4, and (3) resistant to movement artifact. An example of the overall design of the cap can be demonstrated in Figure 2.
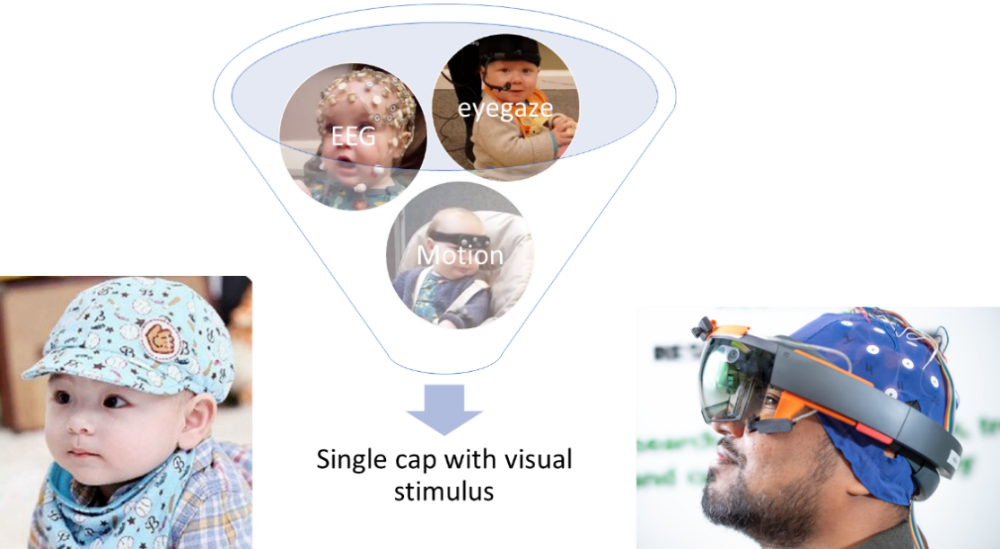
Figure 1.
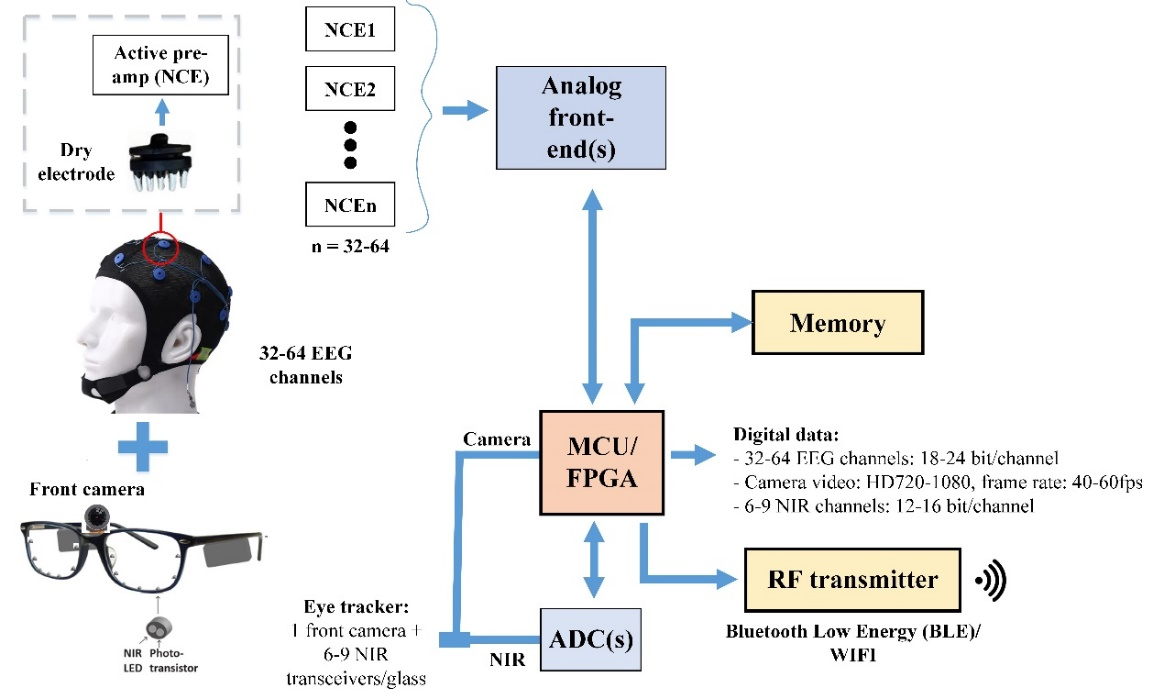
Figure 2.
- Motion-Compensated Neuroimaging using MRI: An Enabling Technology for Novel Neuro-Cognitive-Behavioural Assessments in Children with Cerebral Palsy (*ƯU TIÊN)
- Backgrounds
Magnetic resonance imaging (MRI) is vital to clinical care and research in cerebral palsy (CP), providing a personalised window into neuro-anatomical causes, cognitive and motor outcomes, relevant interventions, and the impact of therapies. However, the MRI experience can be a very difficult one for children because of the intimidating scanner design, loud noises and claustrophobic environment. These aspects exacerbate a fundamental limitation of MRI: motion.
MRI requires subjects to remain still during acquisitions lasting several minutes to an hour. This requirement is violated in every scan to a greater or lesser degree and often leads to image artifacts. Movement results in approximately 20-30% of paediatric scans being nondiagnostic and successful scanning may require children to be still >90% of the time. Higher failure rates occur for clinical groups, particularly if part of the clinical problem is excessive movements as in dyskinetic CP, and higher rates again exist for children aged 1-6 yr.
General anaesthesia (GA) or sedation are frequently used to minimise motion in children undergoing MRI, especially those <7 yr. Although sedation improves the success rate of diagnostic MRI scans compared to non-sedated, the failure rate remains high and the risk of airway obstruction or loss of vital reflex requires careful management.
Therefore, in this research, we propose a different philosophy: allow motion and correct for it, to bring the option and benefits of non-sedated MRI to children, caregivers and practitioners. Prospective motion correction involves using continuous, non-invasive measurements of the subject’s head pose during a scan to constantly adjust the scan sequence (i.e. the magnetic field gradients and radiofrequency pulses). From the scanner perspective, the head does not appear to move at all and we can reconstruct images with minimal artifact. Since 2016, we have been developing prospective motion correction capability for MRI in collaboration with our industrial partner Hobbitview Inc. and now have a benchtop-ready MRI-compatible motion correction system supported by extensive proof-of-concept and preliminary data.
The vision of this project is to merge a motion-corrected neuroimaging paradigm for children with CP – useful in its own right – with eye gaze tracking capability. There are two important drivers for this pairing. First, approximately 50% of children with CP are non-verbal and therefore have no obvious means of communicating with imaging staff or caregivers during scanning. Providing eye-gaze technology to non-verbal children for communication during neuroimaging will ensure a less anxiety provoking experience. Second, eye-gaze behaviour provides a window into cognition and social development, even from early infancy. This opens up the exciting possibility of using eye-gaze responses during functional MRI (fMRI) studies to visualise and quantify regional, time-varying changes in the brain, and to monitor such changes over time. This powerful paradigm is not currently used or available for trans-diagnostic neurodevelopmental groups.
- Proposal
The overarching goal of this research is to make it far easier for children with CP to have successful neuroimaging studies without anaesthesia and to magnify the value of such studies. This will be realised through an enabling technology that simultaneously respects the importance of motion correction in MRI of awake. The 3 specific aims of the project are:
- To develop our current MRI-compatible motion measurement system from benchtop readiness to clinical readiness, and to combine it with the motion correction technology previously developed by our collaborators, Hobbitview Inc. The outcome of this will be a complete motion compensation solution that maximises the value of structural and functional MRI studies and allows them to be performed reliably in the presence of head movement;
- To validate the technology in controls and children with CP. The outcome of this will be a preliminary evaluation of the performance and suitability of the technologies for making measurements in the brain, enabling a new class of experiments, and making non-sedated MRI accessible to CP and trans-diagnostic neurodevelopmental groups.
The project methodology has two distinct phases:
- Phase 1 (2 yr) will address Aim 1 by translating our MRI-compatible head motion tracking method developed at the University of Sydney from a benchtop-ready system (current status) to full integration on a clinical Siemens or GE 3T MRI. This requires computational acceleration of the motion tracking using a graphics processing unit (feasibility of this was demonstrated in [20]) and then interfacing our motion measurements with the proprietary MRI field gradient and RF updating scheme for motion correction developed by our collaborators, Hobbitview Inc. The risk associated with Phase 1 is low since we have strong preliminary data supporting it [19, 20]. Finally, we will validate the integrated prospective motion correction in experiments involving manually-manipulated phantoms and in 10 human volunteers (adults and children) moving under instruction.
- Phase 2 (1 yr) will address Aim 2 and is a pilot validation of the enabling technology developed in Aims 1. We will recruit 45 children with CP (age and gender matched) into three age groups (pre schoolers 3-5 yr, middle childhood 6-8 yr, middle childhood 9-11 yr) with a minimum of 15 children/group. All children will undergo awake neuroimaging sessions with eye-gaze, including both anatomical and fMRI sequences. Children/caregivers and clinical staff (radiographers, neurologists) will complete an experience survey assessing the pre-imaging, imaging, and post-imaging experience using a 5-point Likert scale. We will analyse group differences between the test cohort and controls, where controls represent survey responses from CP families and clinical staff who have only experienced neuroimaging under GA/sedation. Image quality analysis will be performed for all studies to evaluate the degree to which motion-corrected studies are impacted by residual motion artifacts, and the percentage of successful diagnostic MRI sequences. Finally, the fMRI data will allow us to test the feasibility of activation studies based on eye-gaze tasks, analysing group statistics and within-individual comparisons between task and baseline.
